Research
Microcompartments | Protein Secretion | Virus-Like Particles | Protein-based Biomaterials
The Tullman-Ercek lab builds biomolecular devices for a wide range of applications, from medicine (e.g., vaccines, therapeutics, drug delivery, probiotics) to the environment (e.g., bioremediation, sustainable biochemical production, degradable plastics, next-generation biomaterials). We focus on engineering highly organized self-assembling protein systems – the nanoscale building blocks of biology. Such assemblies govern critical processes at all levels of life, from formation of viral capsids to oxygen transport in blood. We investigate:
(1) What fundamental principles govern the precise nanoscale organization of proteins?
(2) How does organization of biochemical processes enhance their performance?
(3) How can we manipulate these protein assemblies to gain new or enhanced functions?
Our current model systems include protein containers, like the MS2 viral capsid and bacterial organelles (microcompartments), and membrane protein machines such as the type III secretion system.
Microcompartments: Organizing Life for Sustainable Biomanufacturing
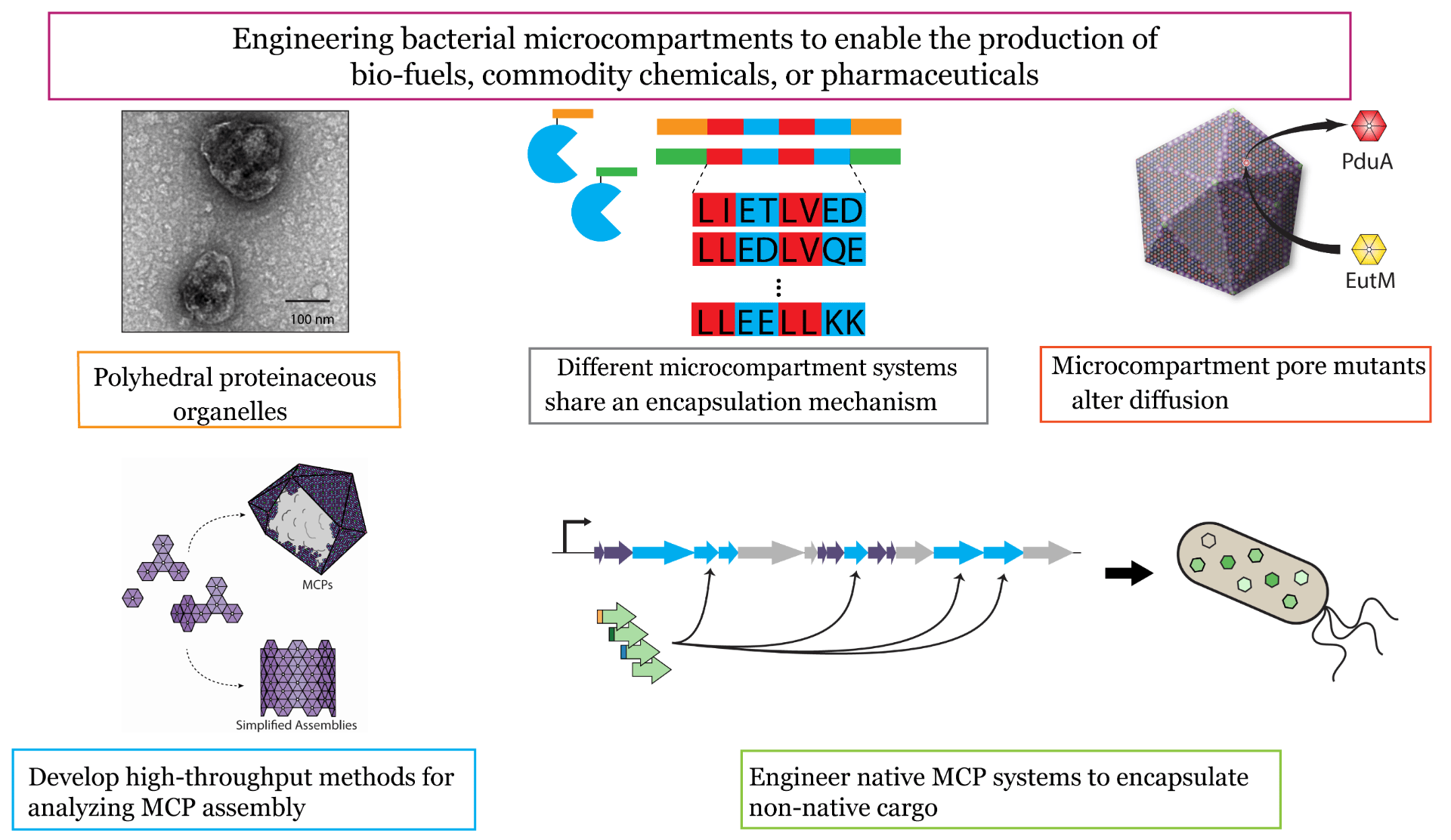
All life is based on segregation of chemical reactions into structures such as organelles, cells, and tissues. We study the benefits and applications of subcellular organization in bacteria, which natively encapsulate some metabolic pathways in proteinaceous organelles called microcompartments. Recently, we have reprogrammed microcompartments to encapsulate enzymatic pathways for production of biofuels and commodity chemicals. In this subgroup, we study the assembly principles of these protein complexes, engineer methods to controllably encapsulate new enzymes, and explore how pathway encapsulation can address metabolic engineering challenges.
Recent Publications:
Mills, C. E., Waltmann, C., Archer, A. G., Kennedy, N. W., Abrahamson, C. H., Jackson, A. D., Roth, E. W., Shirman, S., Jewett, M. C., Mangan, N. M., Olvera De La Cruz, M., Tullman-Ercek, D. Vertex Protein PduN Tunes Encapsulated Pathway Performance by Dictating Bacterial Metabolosome Morphology. (2022). Nature Communications. 13 (3746). DOI: 10.1038/s41467-022-31279-3
Kennedy, N. W., Mills, C. E., Abrahamson, C. H., Archer, A. G., Shirman, S., Jewett, M. C., Mangan, N. M., Tullman-Ercek, D. (2022). Linking the Salmonella Enterica 1,2-Propanediol Utilization Bacterial Microcompartment Shell to the Enzymatic Core via the Shell Protein PduB. Journal of Bacteriology. DOI: 10.1128/JB.00576-21
Kennedy, N. W., Mills, C. E., Nichols, T. M., Abrahamson, C. H., & Tullman-Ercek, D. (2021). Bacterial microcompartments: tiny organelles with big potential. Current Opinion in Microbiology, 63, 36-42. DOI: 10.1016/j.mib.2021.05.010
Li, Y., Kennedy, N. W., Li, S., Mills, C. E., Tullman-Ercek, D., & Olvera de la Cruz, M. (2021). Computational and experimental approaches to controlling bacterial microcompartment assembly. ACS Central Science, 7(4), 658-670. DOI: 10.1021/acscentsci.0c01699
Kennedy, N. W., Ikonomova, S. P., Lee, M. S., Raeder, H. W., & Tullman-Ercek, D. (2021). Self-assembling shell proteins PduA and PduJ have essential and redundant roles in bacterial microcompartment assembly. Journal of Molecular Biology. 433(2), 166721. DOI: 10.1016/j.jmb.2020.11.020
Protein Secretion: Developing Affordable Biomanufacturing
Proteins are used in numerous applications, ranging from detergent stain removers to cancer drugs. However, current methods of commercial protein production are encumbered by long and costly purification processes. Some proteins currently cannot be produced in microbial hosts at all due to cellular toxicity. These problems may be solved by continuously transporting proteins produced in the cell to the extracellular medium, thereby removing the protein from the cell and simultaneously releasing it to an environment with fewer contaminating proteins. This subgroup focuses on harnessing the type III secretion system of bacteria to export proteins of biotechnological interest such as antibodies at high titers. This fascinating membrane protein complex is a nano-scale needle that pumps proteins across the cell boundaries; its natural role is to initiate the infection process. As we make advances in manipulating this system, we also can impact the design of new strategies to combat infections by the gut pathogens that rely on this system.
Recent Publications:
Burdette, L. A., Wong, H. T., Tullman-Ercek, D. (2021). An optimized growth medium for increased recombinant protein secretion titer via the type III secretion system. Microbial Cell Factories, 20(1), 1-13. DOI: 10.1186/s12934-021-01536-z
Burdette, L. A., Leach, S. A., Wong, H. T., Tullman-Ercek, D. (2018). “Developing Gram-negative bacteria for the secretion of heterologous proteins.” Microbial Cell Factories. 17(196). DOI: 10.1186/s12934-018-1041-5
Azam Glasgow, A., Tullman-Ercek, D. (2018). “Type III Secretion Filaments as Templates for Metallic Nanostructure Synthesis.” In: Protein Scaffolds. Methods in Molecular Biology, vol 1798. Humana Press, New York, NY. DOI: 10.1016/j.ymeth.2018.04.014
Azam Glasgow, A., Wong, H. T., Tullman-Ercek, D. (2017). “A Secretion-Amplification Role for Salmonella enterica Translocon Protein SipD.” ACS Synthetic Biology. 6(6). DOI: 10.1021/acssynbio.6b00335
Metcalf, K. J., Tullman-Ercek, D. (2017). “Use of Transcriptional Control to Increase Secretion of Heterologous Proteins in T3S Systems.” In: Type 3 Secretion Systems. Methods in Molecular Biology, vol 1531 (71-79). Humana Press, New York, NY. DOI: 10.1007/978-1-4939-6649-3_6
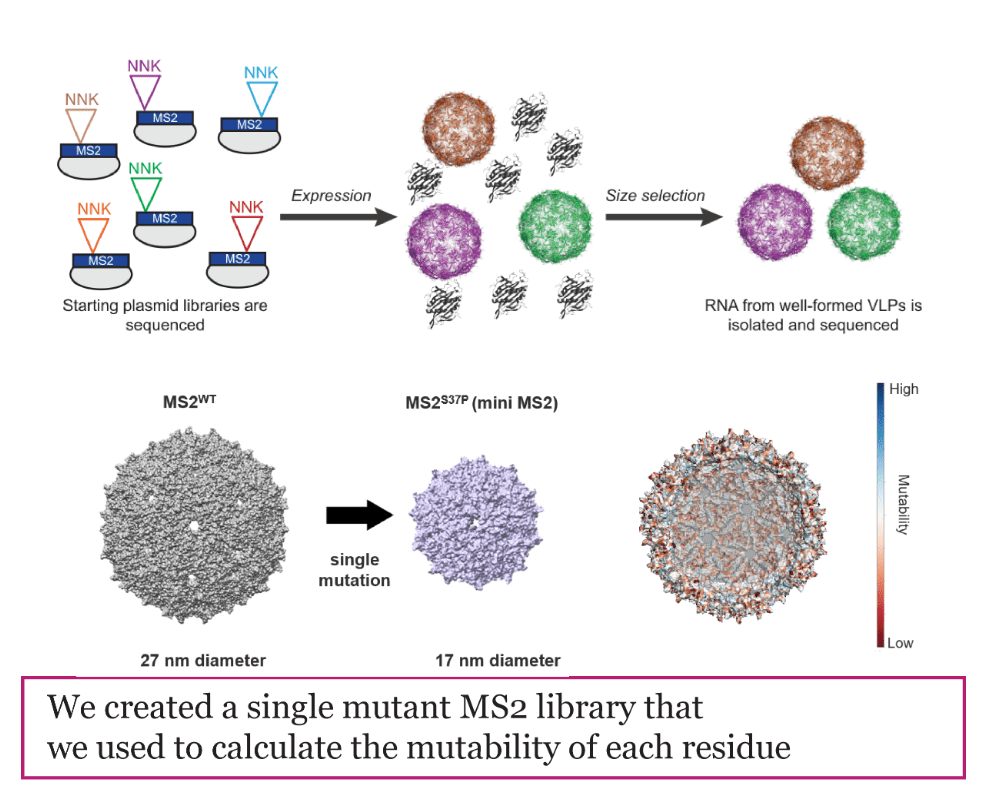
Virus-Like Particles: Vaccines and Drug Delivery
Virus-Like Particles (VLPs) are protein structures derived from viral capsids that protect and deliver cargo to host cells. Our group focuses on adapting VLPs for applications such as targeted drug delivery, gene therapy, vaccines, and imaging diagnostics. However, the capacity to selectively deliver VLPs to target cells in vivo remains a challenge, as immunologic agents clear these macromolecular carriers from circulation before tissue-specific uptake can occur. To overcome these limitations, we study how mutations impact VLP properties, such as size and stability, and use cutting-edge methods including cell-free expression and bioconjugation to “decorate” the VLP shell with new functionality. We collaborate with several groups in academia and industry to test the most promising designs in models relevant to human health.
Recent Publications:
Robinson, S. A., Hartman, E. C., Ikwuagwu, B. C., Francis, M. B., Tullman-Ercek, D. (2020). “Engineering a Virus-like Particle to Display Peptide Insertions Using an Apparent Fitness Landscape.” Biomacromolecules. 21(10): 4194-4204. DOI: 10.1021/acs.biomac.0c00987
Kafader, J. O., Melani, R. D., Durbin, K. R., Ikwuagwu, B., Early, B. P., Fellers, R. T., Beu, S. C., Zabrouskov, V., Makarov, A. A., Maze, J. T., Shinholt, D. L., Yip, P. F., Tullman-Ercek, D., Senko, M. W., Compton, P. D., Kelleher, N. L. (2020). “Multiplexed mass spectrometry of individual ions improves measurement of proteoforms and their complexes.” Nature Methods. 17(4): 391-4. DOI: 10.1038/s41592-020-0764-5
Brauer, D. D., Hartman, E. C., Bader, D. L. V., Merz, Z. N., Tullman-Ercek, D., Francis, M. B. (2019). “Systematic engineering of a protein nanocage for high-yield, site-specific modification.” Journal of the American Chemical Society. 141(9): 3875-84. DOI: 10.1021/jacs.8b10734
Hartman, E. C., Lobba, M. J., Favor, A. H., Robinson, S. A., Francis, M. B., Tullman-Ercek D. (2019). “Experimental evaluation of co-evolution in a self-assembling particle.” Biochemistry. 58(11): 1527-38. DOI: 10.1021/acs.biochem.8b00948
Protein-based Biomaterials: Building Better Materials with Biology
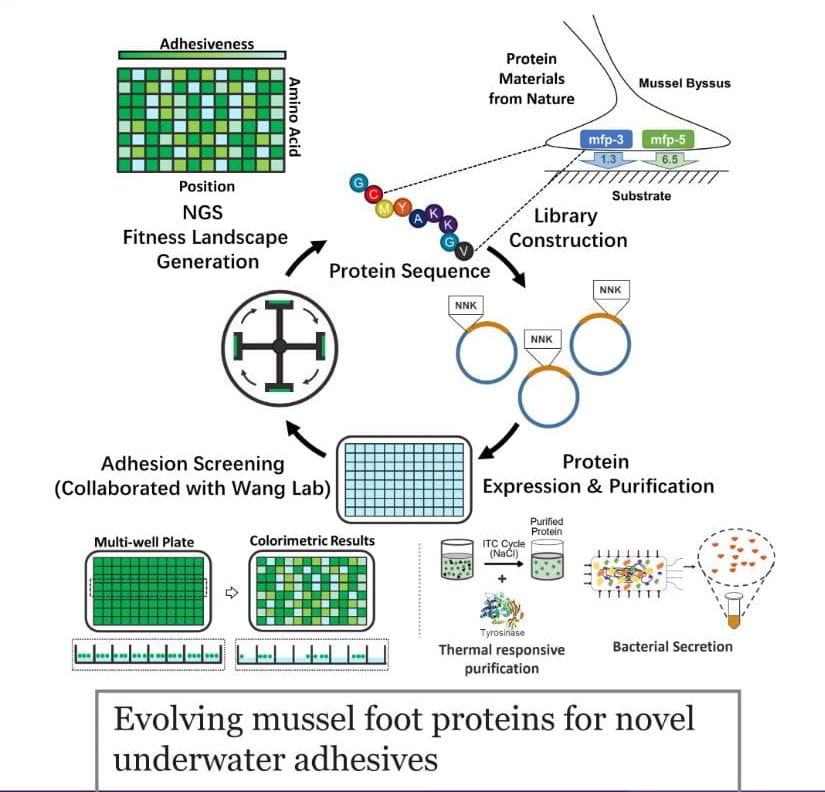
We are using natural biological materials – silk, resilin, elastin, mussel adhesive proteins, and more–as building blocks for engineered materials with user-defined properties. Such goals require understanding the relationship between sequence, structure, and function, and collection of large data sets to inform these design rules. To this end, we collaborate with several labs to develop high-throughput library generation, protein purification, deposition, and property measurement techniques. We envision that this work will eventually permit evolution of novel underwater protein adhesives, new flexible/wearable sensors, and color-changing materials with superior properties.
Recent Publications:
Chen, Y., Wang, Q., Mills, C. E., Kann, J. G., Shull K. R., Tullman-Ercek, D., Wang, M. (2021). High-Throughput Screening Test for Adhesion in Soft Materials Using Centrifugation. ACS Central Science. 7 (7), 1135–1143. DOI: 10.1021/acscentsci.1c00414